
Neither of the unbalanced components in this reaction is present in multiple formulas on the same side of the reaction arrow or is represented in its atomic form. The process described above should be repeated until all of the elements and polyatomic ions that are present in the reaction are balanced. multiply the subscript of each element or polyatomic ion that present in the corresponding chemical formula by the value of the coefficient that was incorporated, in order to update the quantities of those components.write the number that results in the coefficient "blank" on the side of the reaction arrow that has fewer of that component, and.divide the larger quantity of that chemical by the corresponding smaller count,.Once an acceptable component has been selected, start by balancing a chemical that is represented in its atomic form.begin the balancing process by selecting an element or a polyatomic ion that is present in multiple formulas on the same side of the reaction arrow, or.attempt to balance multiple different elements or polyatomic ions with a single coefficient,.When determining the value of a coefficient, do not 
Therefore, one or more coefficients must be written in the "blanks" above, in order to balance this reaction. However, fluorine, F, and bromine, Br, are not balanced, because each of these elements is present in different quantities on the reactant and product sides of the reaction arrow. Since both sides of the reaction contain equal amounts of lithium, Li, this element is balanced. In order for a component of a reaction to be balanced, the reactants and products of the reaction must contain identical total quantities of that type of element or polyatomic ion. The quantities of fluorine, F, lithium, Li, and bromine, Br, that are present in the given equation are summarized in the table that is shown below. Therefore, only individual elements must be considered for this reaction. 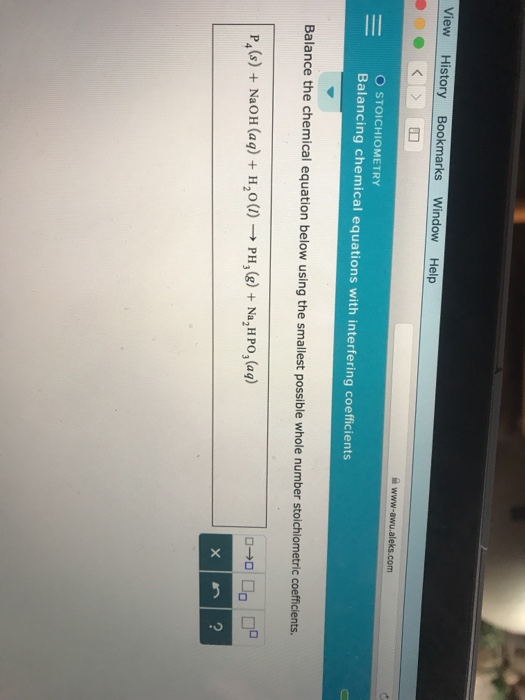
No polyatomic ions are present in any of the formulas that are shown above. In order to balance a chemical equation, the quantities of each type of element and polyatomic ion that are present in the reactants and the products of the reaction must be determined.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
